University of Queensland researchers have found a potential link between inflammation and metabolic dysfunction and treatment-resistant depression. Their preclinical study will inform future clinical trials, contributing to the ongoing development of more personalised treatments for people with mental illnesses.
Associate Professor Susannah Tye and her lab at UQ’s Queensland Brain Institute focus on understanding the biology of treatment-resistant depression, particularly the potential to use key biomarkers to improve diagnosis and treatment. Dr Roger Varela is a postdoctoral research fellow in the Tye lab and led this study.

Dr Varela, what is treatment-resistant depression, and why do you study it?
Treatment-resistant depression (TRD) is not alleviated or able to be adequately managed by classical antidepressant medication. In the lab, we investigate various aspects of TRD, including its causes, which is my area of interest.
Approximately 10-30% of people with depression don’t respond to typical antidepressant drugs, and another 30-40% experience only partial remission. Some people trial several medications, different dosages and combinations of medications over many months or years before doctors can establish that they have TRD.
If we can improve our understanding of TRD’s underlying causes, we can help develop more effective tools for its diagnosis and new treatments to alleviate the personal and social burden associated with this condition.
Why have you chosen to explore inflammation and metabolic dysfunction?
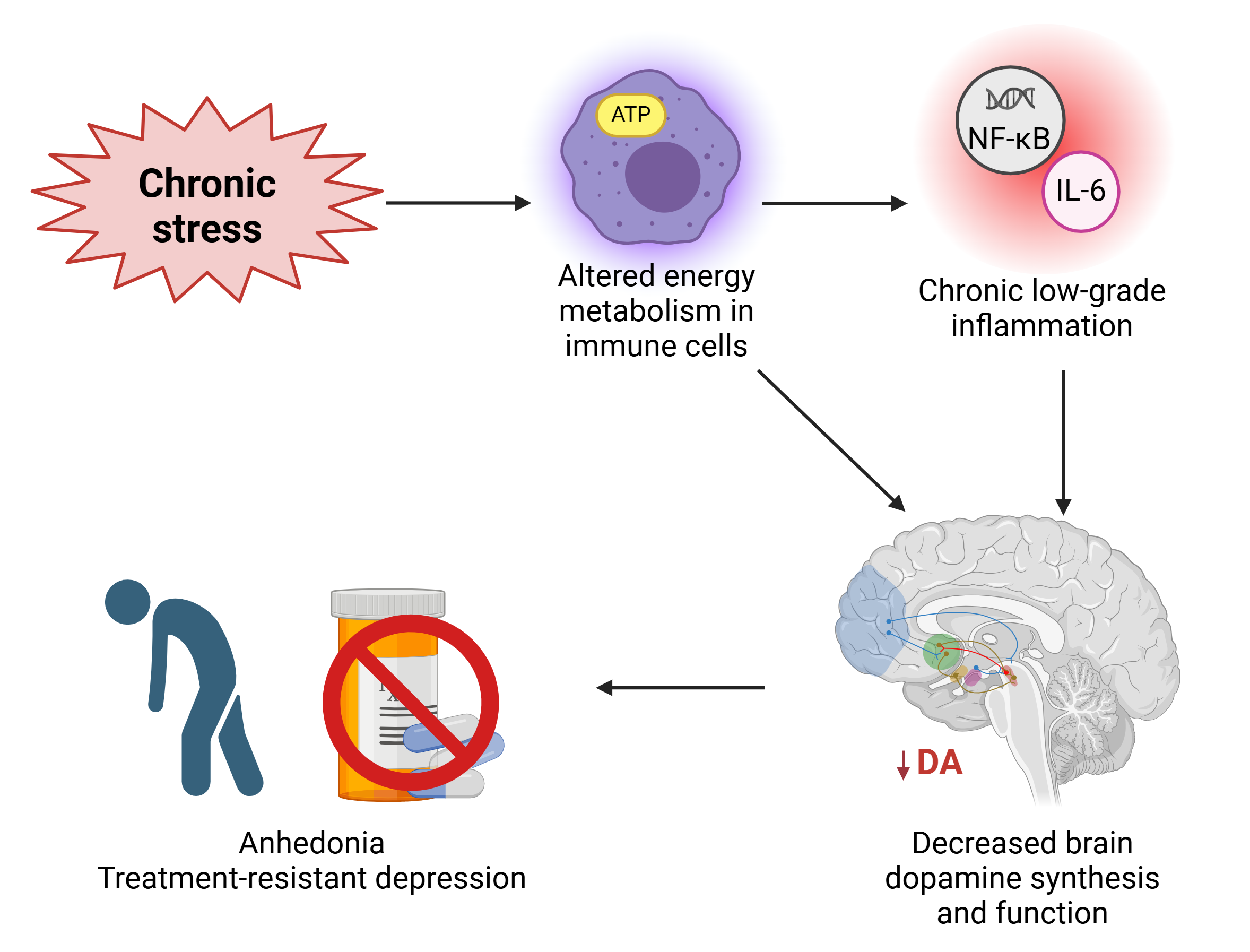
There is evidence in clinical trials that inflammation and metabolic dysfunction are related to depressive symptoms, but it is difficult to measure this link between the body and brain in people. We decided to start with an animal model (rats) to assess how stress, inflammation and the bioenergetics in our immune cells link to what’s happening in the brain. Ideally, we wanted to create a technique that we can translate for use with patient samples and then in clinical trials.
Our study explored how inflammation and metabolic dysfunction induced by chronic stress impair dopamine neurotransmission in the brain, potentially contributing to TRD.
We started by considering inflammation markers linked with dopamine, the ‘feel good’ neurotransmitter and that plays a role in the brain’s reward system and helps to regulate motivated behaviours. We also looked at bioenergetic markers as both inflammation and stress demand a lot of energy. We were excited to find that inflammation and bioenergetic dysfunction were closely correlated with changes in dopamine and motivated behaviour.
What did you find?
In our study, we showed that inflammation and metabolic dysfunction in the body can work as a signal to disrupt dopamine synthesis in the brain, shutting down the reward system and leading to depressive symptoms like anhedonia – the inability to experience joy or pleasure – often seen in TRD and associated with a poor antidepressant response.
This work reinforces the concept of the body-mind connection, i.e. by managing inflammation levels well through diet, exercise, sleep and a positive environment, we can also boost their protective effects on the brain.
Our findings highlight the potential for immune and metabolic markers to offer new paths for diagnosis and personalised treatment for people with TRD. If we can gather a patient’s bioenergetic profile, we may be able to predict whether they will respond or not to a particular antidepressant.
What’s the next step for this research?
This work was conducted with an animal model, but we have several collaborations with clinical TRD research groups. The next step is to use samples collected in research clinics to confirm this paradigm in human subjects. We have already begun this work assessing mitochondrial activity and other markers before and after treatment in these samples.
This knowledge is informing our development of a blood test that can predict a person’s likely response to different antidepressants, which may streamline their diagnosis, improve their treatment and save time and healthcare costs.
Dr Varela, you are from Brazil. How did you come to research TRD in Queensland?
During my PhD in Brazil, I was granted what’s called a “sandwich PhD” fellowship to do part of my training overseas. I did a year of PhD research training at the Mayo Clinic in 2017 under Sue Tye’s supervision, who directed the Translational Neuroscience Laboratory there between 2012 and 2017. I returned to Brazil to complete my doctorate and became aware of a postdoc position in Sue’s lab here at QBI, where she has been since leaving Mayo. I was recruited to her team and moved to Queensland in 2019, where I’ve worked ever since.
Have you seen the interest in your research grow in recent years?
Yes, I think the awareness about the prevalence of depression and other mental health conditions has been increasing in the last few years, especially after the Covid-19 lockdowns. People now understand the severity of these brain diseases and their underlying biological causes. Since the onset of the COVID-19 pandemic in 2020, there has been a noticeable surge in public interest in mental health research, particularly in understanding the role of inflammation and metabolic brain health.
We are excited to advance this important work in collaboration with leading Australian and international partners, supported by funding from the NHMRC and ARC and generous contributions from families and foundations committed to driving innovation in mental health through research and development.
To truly transform outcomes for those affected by mental illness, we need sustained support. With your help, we can accelerate discoveries that offer hope for healing and resilience.
This research was published in Brain, Behavior and Immunity.



